Dual antiplatelet therapy and non-cardiac surgery: evolving issues and anesthetic implications
Article information
Abstract
Dual antiplatelet therapy (DAPT) consisting of aspirin plus a P2Y12 inhibitor (clopidogrel, prasugrel, or ticagrelor) is imperative for the treatment of acute coronary syndrome, particularly during the re-endothelialization period after percutaneous coronary intervention (PCI). When patients undergo surgery during this period, the consequences of stent thrombosis are far more serious than those of bleeding complications, except in cases of intracranial surgery. The recommendations for perioperative DAPT have changed with emerging evidence regarding the improved efficacy of non-first-generation drug (everolimus, zotarolimus)-eluting stents (DES). The mandatory interval of 1 year for elective surgery after DES implantation was shortened to 6 months (3 months if surgery cannot be further delayed). After this period, it is generally recommended that the P2Y12 inhibitor be stopped for the amount of time necessary for platelet function recovery (clopidogrel 5–7 days, prasugrel 7–10 days, ticagrelor 3–5 days), and that aspirin be continued during the perioperative period. In emergent or urgent surgeries that cannot be delayed beyond the recommended period after PCI, proceeding to surgery with continued DAPT should be considered. For intracranial procedures or other selected surgeries in which increased bleeding risk may also be fatal, cessation of DAPT (possibly with continuation or minimized interruption [3–4 days] of aspirin) with bridge therapy using short-acting, reversible intravenous antiplatelet agents such as cangrelor (P2Y12 inhibitor) or glycoprotein IIb/IIIa inhibitors (tirofiban, eptifibatide) may be contemplated. Such a critical decision should be individually tailored based on consensus among the anesthesiologist, cardiologist, surgeon, and patient to minimize both ischemic and bleeding risks.
Introduction
Thrombus formation due to platelet adherence to ruptured plaques plays a pivotal role in the pathogenesis of acute coronary syndrome (ACS) and myocardial infarction [1]. Moreover, platelet aggregation induces the release of secondary messengers that are responsible for further thrombus formation and vasoconstriction [2]. Accordingly, dual antiplatelet therapy (DAPT) consisting of aspirin and a P2Y12 inhibitor, mainly clopidogrel, has become the cornerstone of medical therapy for ACS [3]. The role of DAPT is even more important during the re-endothelialization period after percutaneous coronary intervention (PCI), during which time thrombotic risk is greatest due to endothelial injury by ballooning or stent coverage [4].
With the unavoidable interference in the coagulation cascade, however, DAPT is accompanied by an inevitable increase in bleeding risk, even in patients not exposed to surgical procedures [5]. In this context, surgery not only exposes the patients to additional bleeding risk but also exposes them to an increased risk of thrombosis due to the development of hypercoagulability during the early postoperative period as a result of systemic inflammation [67]. Unfortunately, approximately 5–20% of patients undergoing PCI are presented for non-cardiac surgery within the first 2 years after PCI [8]. Of these, 47% who received a drug-eluting stent (DES) were presented for surgery within 12 months, during which time previous guidelines, based on first-generation DES, mandated the requirement for DAPT [9]. Thus, anesthesiologists are often required to participate in critical decision making regarding the continuation/discontinuation of DAPT. Ideally, the consensus among the anesthesiologist, cardiologist, surgeon, and patient on perioperative DAPT should be individually tailored to minimize both ischemic and bleeding risks.
The most widely used P2Y12 inhibitor, clopidogrel, exhibits variable inter-individual platelet inhibitory responses [10]. Accumulating evidence suggests a close association between high (on-treatment) platelet reactivity (HPR, less inhibition by the drug) and adverse ischemic outcomes [11]. To overcome these limitations, newer P2Y12 inhibitors (prasugrel, ticagrelor) have been developed with improved pharmacodynamic profiles exhibiting more consistent platelet inhibition, although accompanying bleeding risks have also increased [12]. As emerging data show improved ischemic benefits in selected ACS patients treated with prasugrel or ticagrelor versus clopidogrel [1314], anesthesiologists will increasingly encounter these medications. In addition, short-acting, reversible antiplatelet agents have become available in intravenous (IV) forms with potential value as bridge therapy to surgery [1516]. In the context of technical advances in coronary stents, accumulating evidence regarding non-first-generation DES suggests a decreased risk of thrombotic complications compared to first-generation DES, and a reduced necessary duration of DAPT after stent placement [17].
Together with accumulating clinical evidence using the newer P2Y12 inhibitors, prasugrel and ticagrelor, in 2016, the American College of Cardiology (ACC)/American Heart Association (AHA) suggested novel recommendations for perioperative management timing of non-cardiac surgery in patients treated with PCI and DAPT [18]. This review article addresses evolving evidence regarding the abovementioned issues to assist clinicians in making consensus decisions regarding perioperative DAPT in patients undergoing non-cardiac surgery.
Antiplatelet Therapy
While DAPT consists of aspirin and a P2Y12 inhibitor, currently available antiplatelet agents can be broadly classified into five types based on their mechanism of action in hindering platelet aggregation: cyclooxygenase (COX)-1 inhibitor (aspirin), P2Y12 inhibitors (clopidogrel, prasugrel, ticagrelor, cangrelor), glycoprotein (GP) IIb/IIIa inhibitors (abciximab, eptifibatide, tirofiban), phosphodiesterase-III inhibitors (dipyridamole, cilostazol), and protease-activated receptor-1 inhibitor (vorapaxar). The use of cilostazol is mainly limited to patients with peripheral artery disease [19], and only limited data are available regarding the efficacy of triple therapy consisting of DAPT plus cilostazol in patients with coronary artery disease without any added increase in bleeding risk [20]. Similarly, there is emerging but limited evidence regarding the use of vorapaxar in addition to DAPT in patients with coronary artery disease, and ischemic benefits have been reported in patients with previous myocardial infarction or peripheral artery disease, and diabetes [21]. Cilostazol is only a weak, reversible platelet inhibitor with an elimination half-life of approximately 21 h, and thus requires discontinuation for 2 days before surgery [19]. On the other hand, vorapaxar has a long termination half-life of approximately 126–269 h [12]. Nonetheless, with the limited availability of clinical data, the use of cilostazol or vorapaxar will not yet be an issue for ACS patients undergoing surgery, as they will only be used as an adjunct to DAPT on rare occasions. Thus, their pharmacology and anesthetic implications will not be discussed further in this review. The pharmacology of GP IIb/IIIa inhibitors will be briefly discussed with regard to their potential use as a bridge to surgery [15].
Information regarding the elimination half-life of each antiplatelet agent will help in deciding the timing of platelet transfusion in patients presenting for emergent surgery with continuation DAPT (Table 1). In cases of significant perioperative bleeding under such conditions, the logical approach would be transfusion of platelet concentrates. However, if the timing of transfusion falls within the 1–2 elimination half-life of the antiplatelet agent from its last administration, newly transfused platelets will likely be affected by the antiplatelet agent as well, and transfusion may need to be repeated.
COX-1 inhibitor, aspirin
Aspirin was the first antiplatelet agent to show clinical benefits in patients with acute myocardial infarction [22]. Studies also showed that low-dose aspirin treatment led to a consistent risk reduction in vascular events, including myocardial infarction and stroke by 25–30%; therefore, it has become the foundation of antiplatelet therapy for ACS [23]. In conjunction, recommendations or guidelines of various societies mandate the indefinite continuation of aspirin, if possible, when used for secondary prevention (recurrence of cardiovascular disease), particularly in patients with coronary artery disease [1824]. It is noteworthy that aspirin's ability to prevent cardiovascular events for primary prophylaxis (i.e., in the absence of established cardiovascular disease) is unclear without any definitive mortality benefits [23].
Aspirin is an irreversible COX-1 inhibitor that blocks subsequent thromboxane production for the entire lifespan of platelets (usually 7–10 days) [25]. It is able to inhibit 60–70% of platelet function, and is rapidly absorbed after oral ingestion, resulting in a > 90% reduction of thromboxane levels. In addition, aspirin induces non-thromboxane-dependent effects, including enhanced fibrinolysis via acetylation of fibrinogen [26]. In the clinical dose range of 75–100 mg, the elimination half-life of aspirin is approximately 2–4 h. Aspirin inactivates the COX-1 of mature megakaryocytes [27], which are responsible for platelet production in the bone marrow. Thus, COX-1 activity may not return to normal levels for approximately 48 h after discontinuation. Nonetheless, healthy bone marrow is able to replace 10% of the circulating platelets each day. Assuming a normal platelet count, discontinuation of aspirin will lead to the production of more than 30% of healthy unaffected platelets after 3–4 days, which is usually sufficient for normal hemostasis. Indeed, platelet aggregation significantly recovers after 2–3 days of aspirin discontinuation with complete recovery occurring after 5 days [28].
P2Y12 inhibitors, clopidogrel, prasugrel, ticagrelor, cangrelor
DAPT consisting of aspirin plus clopidogrel has become the mainstay of medical therapy for coronary disease after the results of the Clopidogrel in Unstable angina to prevent Recurrent Events trial, which showed a 20% decrease in composite outcome (cardiovascular death, myocardial infarction, stroke) using aspirin plus clopidogrel compared to aspirin plus placebo in patients with ACS [29]. In addition, the ischemic benefits of DAPT were even more defined with regard to reducing the risk of stent thrombosis after PCI [30]. Clopidogrel is a prodrug and therefore requires two metabolic steps to be converted into its active form, which is capable of inhibiting the P2Y12 receptor [1225]. In conjunction, the maximum effects of clopidogrel are achieved after 12–24 h, even after a loading dose of 300–600 mg. The elimination half-life is approximately 4–6 h, although the elimination half-life of its active form is much shorter (0.5–1 h). However, as with aspirin, clopidogrel is able to irreversibly inhibit 60–70% of platelet function and the recovery of platelet function relies on fresh platelet production from megakaryocytes. The recovery of normal platelet function to normal has been demonstrated after 5–7 days of clopidogrel cessation. Because normal hemostasis does not require the complete recovery of platelet function, clinical data regarding surgery have been uneventful after 3–5 days of discontinuation [3132]. Due to its pharmacokinetic properties requiring two-step metabolism, variable inter-individual responses to clopidogrel have been noted, with 4–34% of patients exhibiting inadequate platelet inhibitory response (i.e., HPR), which is related to adverse ischemic outcomes [11].
Similar to clopidogrel, prasugrel requires hepatic activation for conversion to its active form. However, the requirement of prasugrel for a single-step oxidation process is more efficient, yielding 5-fold higher availability of the active metabolite than clopidogrel and inhibiting 90% of platelet function [1225]. This pharmacokinetic advantage allows prasugrel to show a rapid onset of activity at 0.5–4 h, less inter-individual variability, and more consistent platelet inhibition than clopidogrel. Accordingly, accumulating evidence from major clinical trials have demonstrated better ischemic efficacy with prasugrel than clopidogrel in ACS patients undergoing PCI at the expense of increased bleeding risk, which was more serious in patients with a previous history of transient ischemic attack or stroke [13]. Thus, the 2016 ACC/AHA guidelines recommended that prasugrel be favored over clopidogrel in patients with ACS treated with coronary stent but without a history of transient ischemic attack or stroke [18]. The elimination half-life of prasugrel is approximately 7 h, but as with aspirin and clopidogrel, its inhibitory effect is irreversible. Due to its more complete inhibition of platelet function (90%), normalization of platelet function takes longer compared to clopidogrel, requiring 7–10 days after discontinuation [12].
Unlike clopidogrel and prasugrel, ticagrelor directly inhibits the P2Y12 receptor through allosteric modulation [1225]. Therefore, ticagrelor does not require hepatic conversion, but its metabolite is also active accounting for 30–40% of its effects. The unique pharmacokinetic properties of ticagrelor account for its rapid onset of 0.5–4 h, yielding almost complete (90%) inhibition of platelet function. Its clinical efficacy has also been demonstrated in major clinical trials in ACS patients undergoing either PCI or medical therapy, with or without increased bleeding risk compared to clopidogrel [1433]. Thus, the 2016 ACC/AHA guidelines recommended that ticagrelor should be favored over clopidogrel in patients with ACS treated with a coronary stent and in patients with non-ST segment elevation ACS treated with medical therapy alone [18]. The antiplatelet effects of ticagrelor are reversible with an elimination half-life of approximately 7 h requiring twice daily dosing. Complete recovery of platelet function requires 3–5 days of discontinuation [12].
Cangrelor is also an active drug that does not require metabolic activation, and it is the only P2Y12 inhibitor available in IV form [1225]. Because its antiplatelet effect is reversible and short-lived with an elimination half-life of 3–5 min, platelet function returns to normal within 30–60 min of discontinuation, thus requiring continuous infusion. Accordingly, the preoperative administration of cangrelor will not likely pose any problems related to bleeding in surgical settings. To date, its ischemic benefits have been demonstrated in limited clinical studies as an adjunct to DAPT [34]. However, its distinct pharmacokinetic properties make cangrelor a promising agent for bridge therapy before surgery.
GP IIb/IIIa inhibitors, abciximab, eptifibatide, tirofiban
GP IIb/IIIa inhibitors are also available in IV forms and target the final common pathway of platelet aggregation [12]. Therefore, they constitute the most potent form of antiplatelet agent with potential for an unacceptably high risk of bleeding when surgery is performed under their influence. The onset of these agents is rapid, and the elimination half-life of abciximab is 10–30 min, whereas those of eptifibatide and tirofiban are 2–2.5 h. However, the time of return to platelet function after discontinuation is approximately 12 h in abciximab and 4 h in eptifibatide and tirofiban. Fortunately, their clinical indications are limited to the acute phase of PCI, and no long-term therapy with GP IIb/IIIa has been approved. Therefore, their use should rarely be encountered in non-cardiac surgery, but more likely in the setting of emergent surgical coronary revascularization when it is recommended to be stopped for at least 12 h for abciximab and 2–4 h for eptifibatide and tirofiban. Eptifibatide and tirofiban are also promising as bridging agents before surgery.
Non-first-generation DES and New Guidelines for the Timing of Non-cardiac surgery
In addition to the advent of novel antiplatelet agents, there has also been constant improvement regarding the clinical efficacy of DES. While previous guidelines regarding the duration of DAPT after PCI were mostly based on first-generation sirolimus- or paclitaxel-eluting stents, evidence regarding the use of non-first-generation DES (with elution of everolimus or zotarolimus) has emerged [35], forming the basis for the new 2016 ACC/AHA updated guidelines [18]. Clearly, non-first-generation DES showed better ischemic protection with less thrombogenicity requiring a shorter period for re-endothelialization. Accordingly, the necessary duration of DAPT after DES placement has been shortened in the new guidelines [18]. Previously, the optimal duration of DAPT after DES placement was 1 year and surgery was recommended after this time point. If the risk of delaying surgery was greater than the risk of stent thrombosis, surgery was recommended after 6 months of DES placement [9]. In contrast, the updated 2016 ACC/AHA guidelines recommended elective non-cardiac surgery to be performed 6 months after DES placement, and if the risk of delaying surgery was greater than the risk of stent thrombosis, surgery could be considered 3 months after DES placement [18]. Currently, studies addressing the efficacies of most recently developed bioabsorbable stents, and polymer-free and carrier-free drug-coated stents are being performed that may further change the perioperative DAPT management.
Bleeding Risk Versus Ischemic Risk and Their Consequences
Antiplatelet agents interfere with normal platelet function. Therefore, their chronic use is inevitably associated with spontaneous bleeding risk. Since the adoption of a lower dosing regimen (70–100 mg), spontaneous bleeding risk of aspirin has been significantly reduced. Yet, the addition of clopidogrel or ticagrelor greatly increased the bleeding risk, which is highest with the use of prasugrel [1213]. In the case of surgery, even aspirin monotherapy was associated with a modest increase in blood loss, exhibiting a 1.5-fold higher bleeding rate [36]. However, the increase in bleeding risk was not associated with increased morbidity or mortality, except in intracranial surgery [37]. The results of the PeriOperative ISchemic Evaluation 2, aspirin arm (POISE-2, ASA) trial, which was conducted in patients undergoing noncardiac surgery, should be mentioned as it showed increased risk of major bleeding related to aspirin monotherapy in non-cardiac surgery without any increase in the risk of adverse outcome, while no ischemic benefit was observed [38]. However, only 23% of the patients included in the trial had a history of coronary artery disease, and 4.7% received PCI. In this study, aspirin was randomized to patients at risk for vascular complications and not to patients receiving aspirin specifically for secondary prevention of known cardiovascular disease.
Although evidence regarding the interruption of aspirin therapy administered for secondary prevention is mostly limited to observational studies, a meta-analysis involving 50279 patients receiving aspirin for this purpose revealed a few important results that should be taken into consideration when tailoring perioperative DAPT [39]. First, the cardiac complication rate was increased by 3-fold after aspirin withdrawal, which was unacceptably higher (90-fold increase) in patients who received coronary stents. Second, the average time interval from aspirin cessation to coronary events was 8.5 days [3639]. These results and those of other case reports suggest that aspirin therapy for secondary prevention of cardiovascular events should be indefinite, whenever possible, and extreme caution should be exercised when it is stopped for more than 8 days. Concerns have also been raised with regard to rebound hypercoagulability after aspirin withdrawal by excessive thromboxane A2 activity and decreased fibrinolysis, all of which could be further aggravated by surgery-related acute phase reaction [6]. Therefore, the perioperative course may not allow for a time interval of 8.5 days.
This could be more problematic with DAPT, particularly after PCI. Current evidence regarding the bleeding risk of DAPT stems mostly from cardiac surgery and is limited to small numbers of studies or case series in non-cardiac surgery [40]. Overall, the increase in bleeding associated with DAPT in cardiac surgery seems to be 30–50% with increased morbidity (reoperation rate) [41]. However, when non-cardiac surgery is performed during the early re-endothelialization phase after PCI, the rate of myocardial infarction increased steeply to an average of 30% reaching a high mortality of 20–86% [4243]. Accounting for these results, the overall clinical consequences of DAPT cessation during the early re-endothelialization phase after PCI seem to be far more serious than the bleeding risk it accompanies.
Role of Platelet Function Tests and the Search for an Optimal Therapeutic Window
As mentioned above, clopidogrel, currently the main P2Y12 inhibitor used as a DAPT, exhibits considerable inter-individual variability in the platelet inhibitory response [10]. In conjunction, many observational studies in the PCI setting reported robust correlations between HPR measured by various platelet function tests and adverse ischemic outcomes [11]. However, evidence regarding the opposite end of platelet reactivity (i.e., low platelet reactivity) and its association with increased risk of bleeding is limited. Moreover, there have been no randomized, controlled trials showing any clinical improvement upon modifying the antiplatelet regimen according to the results of platelet function tests or genetic testing [18]. In the surgical setting, evidence regarding the use of platelet function tests is mostly limited to cardiac surgeries. Accumulating evidence suggests the usefulness of various platelet function tests in predicting blood loss related to DAPT [4445]. Overall, the preoperative platelet function tests seem to have higher negative predictive value than positive predictive value, meaning that patients found to have normal platelet function (or HPR to P2Y12 inhibitors) do not exhibit increased bleeding risk compared to those not exposed to DAPT [46]. This observation seems logical as perioperative bleeding stems from multifactorial factors. In conjunction, the use of platelet function tests has been incorporated into recent guidelines on the use of antiplatelet agents in cardiac and non-cardiac surgery as a class IIb recommendation [46].
Notwithstanding the potential importance of platelet function tests, several limitations must be taken into consideration in their clinical application. First, there are numerous platelet function tests (PFA-100, VerifyNow, TEG-PM, Multiplate, and so forth) with various and different cut-off values. Second, no platelet function test can provide an optimal therapeutic window of platelet reactivity to P2Y12 inhibitors associated with minimal bleeding as well as ischemic risk. Third, evidence to recommend platelet function tests in non-cardiac surgeries is scarce. At present, the role of platelet function tests in the perioperative period is limited to determining the optimal safe timing of surgery without increasing bleeding risk related to DAPT by demonstration of normal platelet function (or HPR) or guiding platelet transfusion in bleeding patients [4647]. The search to overcome the abovementioned limitations should be continued to enable tailored perioperative DAPT aimed at providing an optimal therapeutic window based on clear-cut criteria.
Potential bridge Therapy to Surgery
The rationale and necessity of bridge therapy to prevent stent thrombosis without the added burden of increased perioperative blood loss is indisputable. Safe bridge therapy has become feasible with the advent of different classes of rapid-onset, short-acting, reversible, and IV forms of antiplatelet agents. Without solid evidence, heparin (either unfractionated or low-molecular-weight) has been suggested as a bridge; however, arterial thrombosis is more dependent on the activation of platelets than the coagulation cascade [1]. Moreover, concerns have been raised regarding the potential harm of unfractionated heparin exposing the platelets to activation by adenosine diphosphate and actually augmenting thrombosis formation [48].
Although few in number, clinical studies using a GP IIb/IIIa inhibitor (tirofiban, eptifibatide) as a bridge has shown promising results [15], while another study in 67 patients undergoing mostly non-cardiac surgery indicated acute ST changes in two patients occurring in the immediate postoperative period [49]. With regard to cangrelor, a recent bridging antiplatelet therapy with cangrelor in patients undergoing cardiac surgery trial involving 210 patients with ACS or recent PCI undergoing surgical coronary revascularization showed consistent maintenance of platelet inhibition with cangrelor without any increase in major bleeding events [16]. More recently, a systematic review of clinical trials and case series of bridge therapy involving 420 patients depicted overall freedom from ischemic events and bleeding/transfusion in 96.2% and 72.6% of the patients, respectively [50]. The median period after PCI (mostly DES placement) upon surgery ranged from 2 to 14 months, with most of the studies showing a period of 2.6 months. Thus, although both agents showed promise, additional evidence regarding the clinical efficacy of both GP IIb/IIIa inhibitors and cangrelor as a bridge for discontinuation of a P2Y12 inhibitor in patients at risk for stent thrombosis is required. Of note, only clopidogrel was discontinued and aspirin was continued in all of the clinical trials addressing the efficacy of bridge therapy.
Recommendations on Perioperative DAPT Management for Non-cardiac Surgery
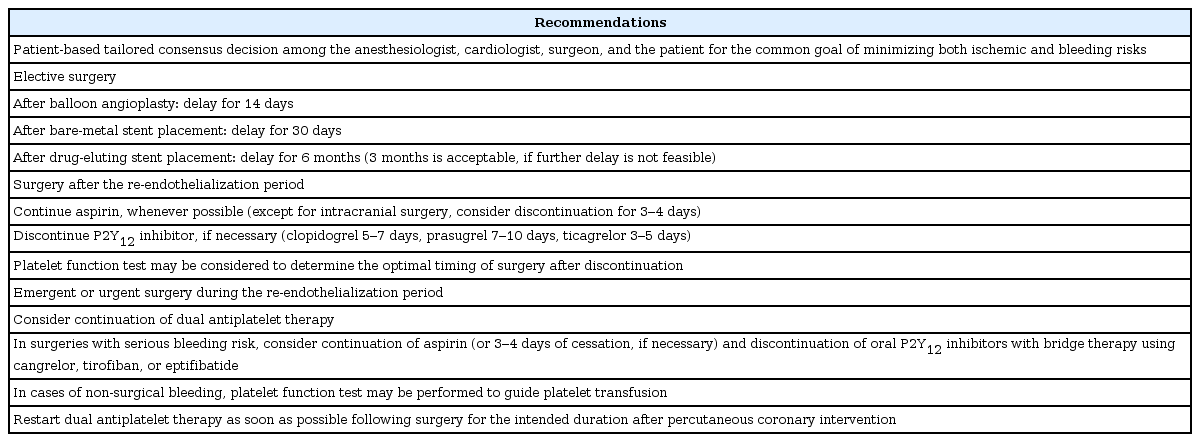
Based on current evidence, it seems imperative to delay surgery after PCI during the re-endothelialization period, which would be 14 days after ballooning, 30 days after bare metal stent placement, and at least 3 months (preferentially 6 months) after DES placement [18]. After this period, it is generally recommended that the P2Y12 inhibitor be stopped for the necessary duration to allow platelet function recovery (clopidogrel, 5–7 days; prasugrel, 7–10 days; ticagrelor, 3–5 days) and that aspirin be continued, except for intracranial procedures when aspirin cessation may be considered for 3–4 days prior to surgery. The duration of P2Y12 inhibitor discontinuation may also be guided by platelet function tests, which may be helpful in avoiding unnecessary delay of surgery by confirming platelet functional recovery regardless of the discontinuation date, particularly with clopidogrel. In emergent or urgent surgeries that cannot be delayed beyond the recommended period after PCI, proceeding to surgery with continued DAPT should be considered as the risk and clinical consequences of stent thrombosis are far greater than the risk of bleeding. For intracranial procedures or other selected surgeries in which increased bleeding risk may also be fatal, cessation of DAPT (possibly with continuation or minimized interruption [3–4 days] of aspirin) with potential bridge therapy using cangrelor or GP IIb/IIIa inhibitors may be contemplated. In cases of non-surgical bleeding with continuation of aspirin or DAPT, platelet function test may be performed to guide transfusion of platelet concentrates. Regardless of the elapsed time of surgery after PCI, DAPT should be restarted in the postoperative period as soon as possible for the originally intended duration. It is important to make a consensus regarding perioperative DAPT among the anesthesiologist, cardiologist, surgeon, and patient as well for the common goal of minimizing both ischemic and bleeding risks (Table 2).
Conclusions
Technical advances related to coronary stents and antiplatelet agents are continuously evolving with the reporting of relevant clinical evidence. While there are numerous guidelines, many challenging clinical situations require a consensus decision based on the expert opinions of the attending physicians. Therefore, a comprehensive understanding regarding the pharmacology of novel antiplatelet agents and their clinical applicability, the clinical efficacies of the newer coronary stents and their risk of stent thrombosis, and the role of platelet function tests must be determined. Incorporating such information, the goal of perioperative management of DAPT in patients with ACS, treated either with or without PCI, should be set to provide optimal tailored management.