Cardioprotection and ageing
Article information
Abstract
With an increase in the elderly population and an increase in the prevalence of age-related cardiovascular disease, anesthesiologists are increasingly being faced with elderly patients with known or suspected ischemic heart disease in the perioperative period. Although early reperfusion remains the best strategy to reduce ischemic injury, reperfusion may damage the myocardium. Adjuvant therapy to revascularization is therefore necessary. To develop better strategies to prevent ischemia-reperfusion injury in older patients, we need to understand the aged myocardium, which has undergone structural and functional changes relative to the normal myocardium, resulting in reduced functional capacity and vulnerability to ischemia-reperfusion injury. In addition, innate or acquired cardioprotection deteriorates with aging. These changes in the aged myocardium might explain why there is poor translation of basic research findings from young animals to older patients. In this review, I discuss changes in intracellular signaling associated with myocardial ageing that have an effect on ischemia-reperfusion injury, and I discuss the efficacy of cardioprotection afforded by ischemic and pharmacologic pre-and post-conditioning in the aged myocardium. Finally, I outline strategies to restore protection in the aged myocardium.
Introduction
The incidence and prevalence of cardiovascular diseases, including ischemic heart disease, are increasing in Korea, most likely due to the ageing of the population and adoption of a Westernized diet. Cardiovascular disease is the leading cause of mortality in the elderly. Two-thirds of myocardial infarctions occur in individuals older than 65 years-of-age, and 80% of deaths caused by myocardial infarction are in elderly patients. These data indicate that ischemic heart disease is prevalent in the elderly, and that the mortality due to ischemic heart disease increases with ageing. According to the National Statistical Office, individuals aged 65 years or older comprised 9.9% of the Korean population in 2007, and this figure is expected to exceed 14% in 2013, thus Korea is expected to become an 'aged society' in the near future. With the rapid increase in the elderly population, the incidence of ischemic heart disease in these elderly individuals is also expected to increase rapidly. Aggressive therapeutic strategies may be required to treat ischemic heart disease in the elderly in order to improve their qualities of life.
Although reperfusion remains the best strategy for the treatment of ischemic heart disease, reperfusion itself injures the myocardium. A study that targeted patients with acute myocardial infarction showed that mortality is higher and left ventricular dysfunction is greater in patients aged 75 years or older despite successful revascularization with thrombolysis [1]. This study proved that the aged myocardium is sensitive to reperfusion injury. Off-pump coronary artery bypass graft surgery has increased the prevalence of cardiac surgery performed in the elderly. Although off-pump coronary artery bypass graft surgery has several advantages such as less neurologic complications and less inflammatory reactions, ischemia-reperfusion injury is not avoidable because of the elevation of the heart during surgery and transient occlusion for coronary anastomosis. Thus, it is very important to develop strategies to reduce ischemia-reperfusion injuries in the aged myocardium during the perioperative period.
A multitude of studies have investigated strategies to protect the heart against ischemia-reperfusion injury, either by ischemic pre- and post-conditioning or by mimics of pharmacologic stimuli. However, the clinical data are inconsistent. The poor translation of basic research results to clinical application may be due to the characteristics of the target population; although most experimental studies have been performed in healthy young animals, the patients in the operating room are diseased and old. We therefore need to understand the aged myocardium in order to develop strategies to reduce myocardial ischemia-reperfusion injuries in these patients.
In this review, I discuss (1) functional and structural changes in the aged myocardium, especially mitochondrial changes, (2) the increased susceptibility to ischemia-reperfusion injury that is characteristic of the aged myocardium, including the changes in intracellular signaling important to ischemia-reperfusion injury, (3) loss of cardioprotection with ageing, and finally (4) strategies to restore protection in the aged myocardium.
Functional and structural changes in the aged myocardium
Ageing affects cardiomyocytes at different levels. At the level of DNA, mutations and telomere shortening are observed in the aged myocardium. Telomere shortening might be associated with limited regenerative capacity [2] and increased mortality [3]. Based on changes in nuclear and mitochondrial gene expression profiles, protein expression in the aged myocardium is altered and is associated with a shift from fatty acid to carbohydrate metabolism and changes in intracellular matrix and signal transduction components [4]. The handling of cellular waste by autophagy is also altered. Aged cardiomyocytes have decreased tolerance to stress such as ischemia-reperfusion and increased susceptibility to apoptosis and necrosis. Increased apoptosis and necrosis reduce the number of cardiomyocytes and result in the development of hypertrophy in the remaining cardiomyocytes. All of these changes decrease contractile function, resulting in decreased left ventricular systolic and diastolic function and decreased peak cardiac output [5].
Mitochondria are the powerhouses of the cell. Energy supplied by mitochondrial oxidative phosphorylation provides more than 70% of the ATP that the myocardium requires [6]. Mitochondria also control ion homeostasis, generate reactive oxygen species (ROS), and are involved in apoptotic/antiapoptotic signaling. Aged cardiomyocytes are sensitive to oxidative stress, which injures mitochondria and makes them larger. These giant mitochondria are not removed by autophagy, and they accumulate in the cardiomyocytes. These injured mitochondria contain damaged, mutated DNA, resulting in the production of mutant proteins, which accelerate ROS generation in aged cardiomyocytes. In addition to excessive generation of ROS, the antioxidant capacity of cells decreases with aging due to increases in levels of manganese-containing superoxide dismutase and decreases in either catalase or glutathione. Mitochondria also play an important role in endogenous cardioprotection. The opening of the mitochondria permeability transition pore (MPTP) is critical for the initiation of cell death [7]. In aged mitochondria, MPTP opening has been observed to be more sensitive than that in young mitochondria [8].
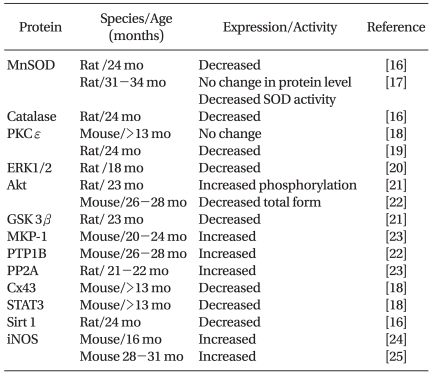
The expression and/or activity of protein kinases, which play a critical role in cardioprotection, are also altered in the aged myocardium (Table 1). The expression level or phosphorylation status of reperfusion injury salvage kinase (RISK) elements, including Akt (protein kinase B), ERK1/2 (extracellular signal-regulated kinase 1/2), and GSK 3β (glycogen synthase kinase 3β), are altered in the aged myocardium. Phosphatases such as protein phosphatase 2A (PP2A) or protein tyrosine phosphatase 1B (PTP1B, which reduce phosphorylation of proteins and suppress their activation, are increased. Levels of Cx43 (connexin 43), STAT3 (signal transducer and activator of transcription 3), and Sirt 1 (sirtuin), which play a role in cardioprotection, are also decreased.
Aged cardiomyocytes are susceptible to calcium overloading [8,9] because of a reduction in the density and activity of calcium regulating proteins such as the Na+/Ca2+ exchanger, Ca2+ ATPase, and ryanodine receptor [10-13]. In addition, modification of the amount and composition of cardiolipin in mitochondria [14] contributes to the breakdown of calcium homeostasis, which is susceptible to the opening of MPTP [15].
Ischemia-reperfusion injury in the aged myocardium
Advanced age is an independent risk factor for poor cardiac outcome. With ageing, comorbidities such as hypertension, atherosclerosis, congestive heart failure, or diabetes may affect the sensitivity of the aged myocardium to ischemia-reperfusion injury. However, several studies have shown that increased mortality in the elderly is independent of the presence of hypertension, diabetes, previous myocardial infarction, number of diseased coronary arteries, and the location of infarction [15,27]. These results indicate that the aged myocardium is vulnerable to ischemia-reperfusion injury.
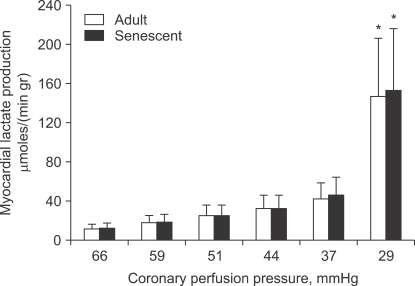
The aged myocardium has decreased tolerance to ischemia-reperfusion injury, though the ischemic threshold is not affected by age (Fig. 1). Decreased tolerance to ischemia-reperfusion injury has been demonstrated in middle-aged mice (12 months); this decreased tolerance became more prominent with ageing (18 months and 24-28 months) [29]. However, the results from a study of the human atrium were inconsistent. Although creatine kinase release after 90 min of ischemia followed by 120 min of reperfusion was not affected by age [30], the recovery of contractile force after 30 min of hypoxia followed by 30 min of reoxygenation was reduced in the atrium of patients aged 60 years or older compared to that of adults younger than 60 years [31].
The increased susceptibility to ischemia-reperfusion injury seen in the aged myocardium might be associated with enhanced oxidative stress. Oxidative damage was found to be aggravated in the aged myocardium of 20-month-old rats [30] and reperfusion with superoxide dismutase and catalase improved contractile function. Furthermore, treatment of the aged myocardium of 24-month-old rats with acetylcarnitine improved the recovery of contractile function and reduced lactated dehydrogenase levels [32].
Loss of protection in the aged myocardium
Ischemic preconditioning (IPC)
Since the first demonstration of ischemic preconditioning in 1986 by Murray et al., IPC has been considered to be one of the strongest forms of endogenous cardioprotection. IPC has two different phases; an early phase that develops within a few minutes and lasts up 2-4 hours and a late phase that develops 6-12 hours and lasts up to 4 days. Complex intracellular signaling pathways mediate IPC, and ROS generated in the mitochondria act as triggers or mediators. Although several effector candidates have been evaluated, the evidence that MPTPs or mitochondrial KATP channels are the effectors is most convincing (Fig. 2). Although simplification of the cascades into a trigger, mediator, and effector can help to clarify the mechanism of IPC, it should be kept in mind that there is crosstalk between the underlying signaling components and that the underlying mechanism is more complex.
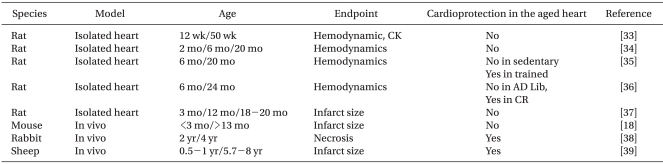
The effect of ageing on the ability of preconditioning to protect hearts from ischemia-reperfusion injury has been studied intensively (Table 2). The majority of studies demonstrated loss of cardioprotection by IPC with ageing. However, IPC was preserved in 2- 4-year-old rabbits [38] and 5.7- to 8-year-old sheep [39]. Based on the maximal life span of these species (Table 3), however, it is unlikely that these animals are suitable models for investigating ageing in humans.
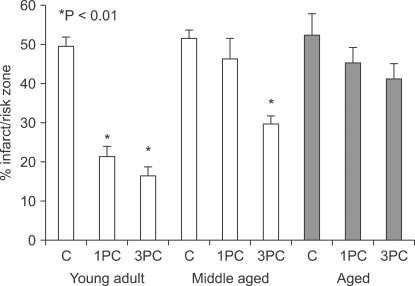
When preconditioning stimuli were increased, cardioprotection was achieved in middle-aged rats. However, cardioprotection induced by IPC was lost in the aged rats, irrespective of the strength of the stimuli (Fig. 3). Indirect data using agonists for receptors or channel openers involved in cardioprotection showed that IPC signaling cascades are impaired in the aged myocardium [37].
IPC has been shown to occur naturally in the form of prodromal angina prior to acute myocardial infarction, reducing the infarct size in patients [41,42]. In clinical studies, the measurement of infarct size is not available as an end-point, and surrogate end-points are used. For examples, ST changes on EKG, biologic markers such as creatine kinase (CK), CK-MB, and troponin-T, and clinical outcomes such as recovery of cardiac functions, length of hospital stay, mortality, and morbidity are frequently used. Clinical studies of IPC during revascularization and prodromal angina within a 48 h prior infarction have consistently demonstrated that IPC is not cardioprotective in elderly patients in terms of recovery of cardiac function, biologic markers, or clinical outcomes [43-45]. However, in vitro studies have showed less consistent results. After IPC in elderly patients, biological markers such as CK were reduced, but contractile function did not improve. Apart from age, comorbidities and medications such as angiotensin-converting enzymes [46,47], COX-2 inhibitors, or sulfonylureas [48] complicate analysis of the IPC effect in the elderly.
Pharmacologic preconditioning
Anesthetic preconditioning (APC) by isoflurane was found to be effective in middle-aged (10-12 months), but not in aged rat hearts (20-24 months) [49,50]. Similarly, in isolated guinea-pig hearts, APC by sevoflurane was attenuated with ageing. Helium preconditioning-induced cardioprotection was not preserved in aged rat hearts [51], and enkephalin preconditioning did not protect aged mouse hearts (24-26 months) [52]. Interestingly, chronic morphine treatment was shown to be cardioprotective via protein kinase A in the aged heart in contrast to loss of acute morphine treatment in the aged heart [53].
Several components of the aged myocardium, included cardioprotection by APC, may be defective. The increase in ROS due to APC is impaired in the aged myocardium, even though the baseline ROS level is high in the aged myocardium [49]. Coupling between the opioid receptor and p38 MAPK (mitogen-activated protein kinase) was shown to be lost in the aged mouse heart, and phosphorylation of the downstream protein HSP27 (Heat shock protein 27) was reduced [52,53].
Ischemic and pharmacologic postconditioning
Interventions performed at reperfusion to protect the myocardium, such as post-conditioning, may be more clinically relevant because it is difficult to anticipate when an ischemic event will occur. IPC is not cardioprotective in aged mice [23]. However, the cardioprotective effect of IPC depends on the stimuli strength in the middle-aged myocardium [54]. The loss of the IPC effect in the aged myocardium was associated with reduced expression and phosphorylation of STAT3 [54,55]. Furthermore, levels of the phosphatase MKP-1 were increased in the aged myocardium, and inhibition of MKP-1 resulted in the restoration of cardioprotection by ischemic postconditioning [23]. ERK, a target protein of MKP-1, was not activated by ischemic postconditioning in the aged myocardium, but was restored in aged hearts treated by a MKP-1 inhibitor [38]. Therefore, in the aged heart, impaired activation of ERK 1/2 might affect downstream components such as STAT3. I found that isoflurane-induced postconditioning was not cardioprotective in old rats (20-24 months) (data not published). The reperfusion salvage kinase pathway, including Akt, its downstream effector GSK3β, and ERK1/2 was not activated by anesthetic postconditioning, which may be associated with the loss of cardioprotection in the aged rat heart. Therefore, the aged myocardium is not protected by pre- or post-conditioning, which is associated with impaired signal transduction pathways.
Restoration of protection in the aged myocardium
Diet
Caloric restriction attenuates cellular ageing and promotes longevity [4,56]. Furthermore, caloric restriction renders cardiomyocytes resistant to ischemic injury. In food-restricted old rats, the cardioprotective effect of ischemic precondition was preserved [36]. Recovery of the developed pressure after ischemia-reperfusion and the occurrence of arrhythmia were evaluated in 6- and 24-month-old rats that had been on a caloric restriction diet since weaning [36,57]. The recovery of developed pressure was partially improved and arrhythmia was reduced in the food-restricted old rats. In a study of short-term caloric restriction, 24-month-old rats were fed 90% of their caloric intake ad libitum for 2 weeks followed by 65% of their caloric intake ad libitum for 2 weeks. The left ventricular pressure was then evaluated in isolated perfused hearts. These authors found that short-term caloric restriction improved tolerance to ischemia, but did not restore IPC-induced cardioprotection [57]. Increased tolerance to ischemia by short-term caloric restriction was related to increased phosphorylation of AMP-activated kinase [58]. Increased levels of adiponectin induced by caloric restriction may contribute to the restoration of the IPC effect in aged hearts [58,59].
Exercise
Exercise contributes to the preservation of endogenous cardioprotection in the aged heart. Elderly rats (24-month-old) were trained using a swimming protocol of 40 min/day, 5 days/week for 6 weeks. Developed pressure in the left ventricle was partially improved by IPC in these trained rats. However, the combination of caloric restriction and exercise restored IPC-based cardioprotection more completely [60]. The mechanisms of exercise-induced cardioprotection or restoration of endogenous cardioprotection are not clear. However, a reduction in mitochondrial ROS formation, increased coronary circulation, increased levels of antioxidants such as manganese superoxide dismutase and catalase, altered expression of mitochondrial and sarcoplasmic KATP channels, and altered gene/protein expression/activation of factors such as HSP and IGF-1 may contribute to exercise-induced cardioprotection [62].
The beneficial effects of exercise on IPC-induced cardioprotection have been demonstrated in elderly patients. High levels of physical activity preserve the cardioprotective effect of prodromal angina in elderly patients [62].
Conclusions
The prevalence of elderly patients with ischemic heart disease is expected to increase as life expectancy and the prevalence of ischemic heart disease increase. We therefore need to understand the characteristics of the aged myocardium and develop strategies to limit ischemia-reperfusion injury in the aged myocardium. Even though the mechanisms of increased susceptibility to ischemia-reperfusion and loss of protection in the aged heart are not fully elucidated, the aged myocardium is known to undergo structural and functional changes associated with impaired signal transduction. These changes may be associated with increased vulnerability to ischemia-reperfusion injury and loss of cardioprotection characteristic of the aged myocardium.
Most experimental studies have evaluated APC in healthy juvenile hearts. Unfortunately, this is far from the clinical reality. Most cardiac events in humans occur later in life, and most patients have comorbidities such as diabetes or hypercholesterolemia. Importantly, aged and diseased hearts behave differently from younger and healthier hearts. Future studies should focus on targeting populations with an aged or diseased myocardium. Furthermore, a more detailed understanding of the molecular mechanisms altered in the aged myocardium is required to identify candidates for pharmacologic manipulation. If cardioprotection is not triggered by ageing or certain drugs, cardioprotection can be provoked by activation of cell surface receptors. However, if activation of receptors or intracellular transduction signaling pathways is impaired, pharmacological agents acting proximal to or at the end-effector might bypass the age-related defects and provide cardioprotection. For example, inhibitors of MPTP might be cardioprotective in the aged myocardium by preventing the opening of the MPTP. However, it is not yet known whether functioning of the MPTP is altered in the aged myocardium, and the function of the MPTP has not been thoroughly elucidated.
Although exercise and caloric restriction might potentially restore endogenous cardioprotection, these are primary prevention strategies rather than strategies adjuvant to revascularization. Stem cell therapy may replace the damaged myocardium or restore endogenous cardioprotection in the aged myocardium [38,63]. However, the differentiation, replication, and long-term stability of stem cells need to be studied further. Research on the mechanisms of endogenous cardioprotection and their alteration with ageing is required to develop effective strategies to reduce ischemia-reperfusion injury in the elderly.